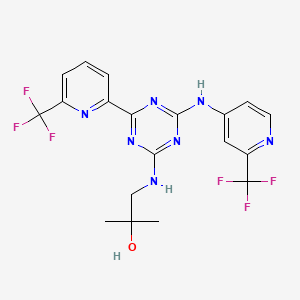
pan-Canadian Oncology Drug Review Final Clinical Guidance Report Enasidenib (Idhifa) for Acute Myeloid Leukemia
IJMS | Free Full-Text | New Perspectives in Treating Acute Myeloid Leukemia: Driving towards a Patient-Tailored Strategy
Appendix A: Drugs Approved in Canada, 2012/13–2018/19, Included in Analysis, with EMA and FDA Equivalents
ROYALTY PHARMA ACQUIRES RIGHTS TO AGIOS' ROYALTY ON IDHIFA® (ENASIDENIB) WORLDWIDE NET SALES AND OUTSTANDING REGULATORY MILES
pan-Canadian Oncology Drug Review Final Clinical Guidance Report Enasidenib (Idhifa) for Acute Myeloid Leukemia

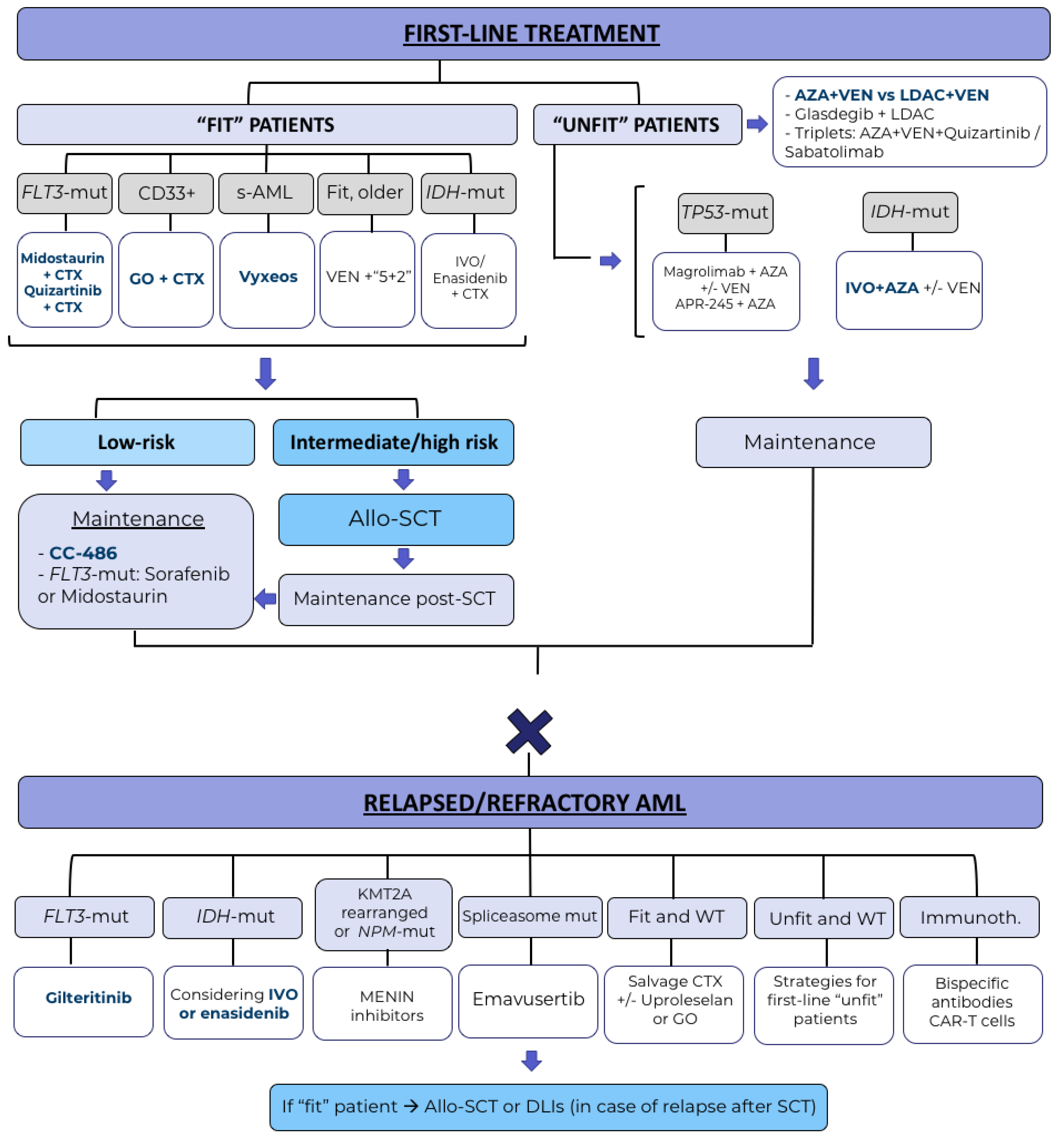
Harnessing the benefits of available targeted therapies in acute myeloid leukaemia - The Lancet Haematology
pan-Canadian Oncology Drug Review Final Clinical Guidance Report Enasidenib (Idhifa) for Acute Myeloid Leukemia
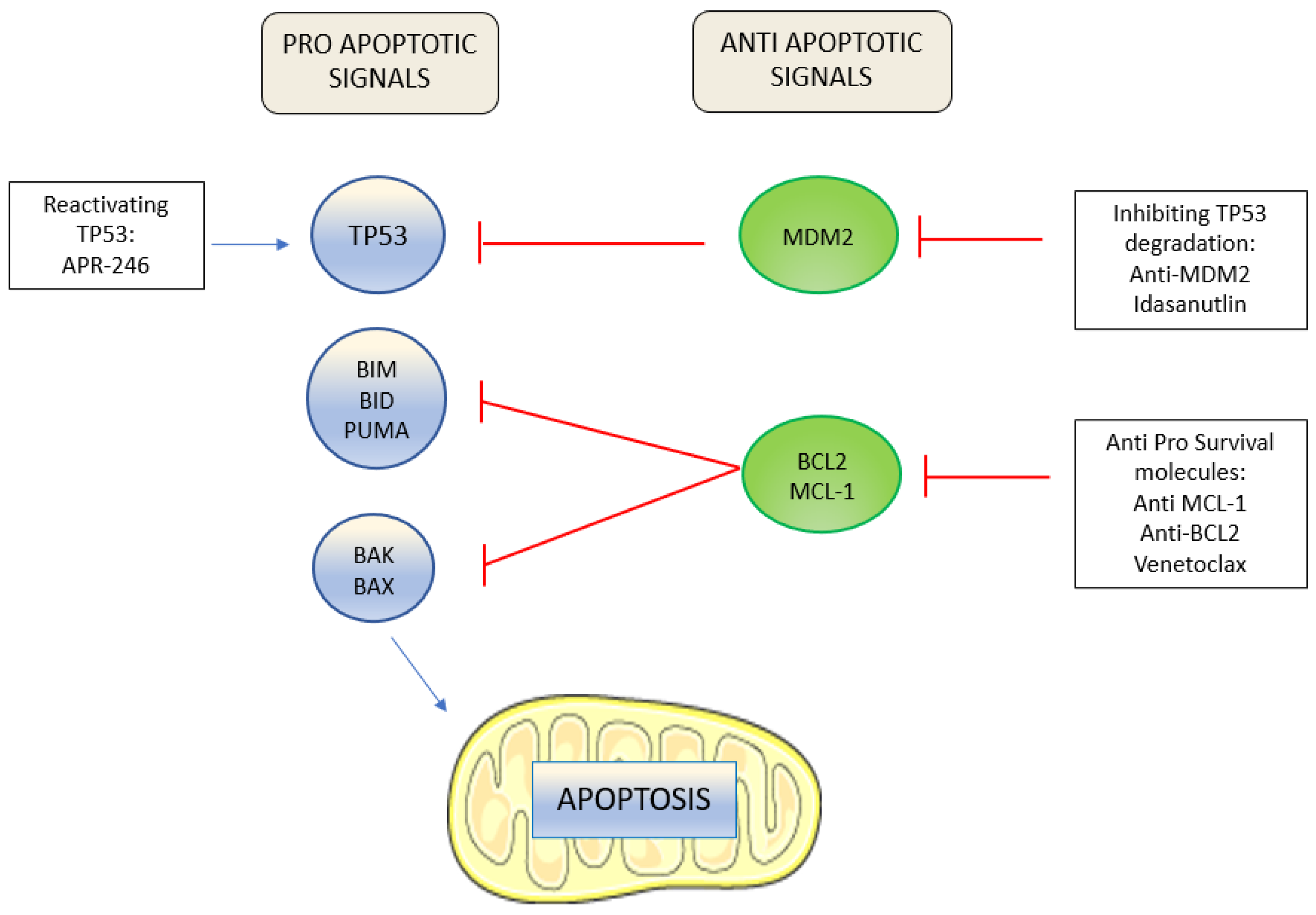
![PDF] Midostaurin, enasidenib, CPX-351, gemtuzumab ozogamicin, and venetoclax bring new hope to AML. | Semantic Scholar PDF] Midostaurin, enasidenib, CPX-351, gemtuzumab ozogamicin, and venetoclax bring new hope to AML. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/349470f78f637dca9405e586f6f86c2659eddcd8/7-Table1-1.png)