Cleaning Validation - Regulations and Guidelines / Toxicology Acceptance Criteria / Examples from the Practice| gempex – THE GMP-EXPERT

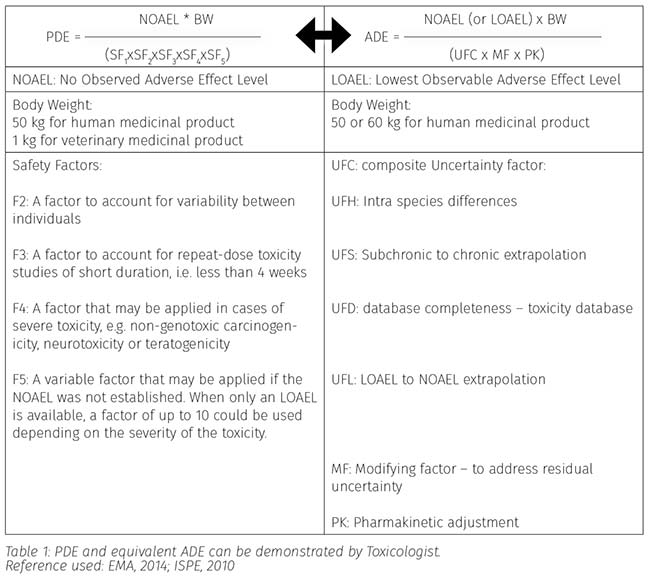
Cleaning validation -Health based safety MACO limit-A Confusing strategy -ADE or PDE and NOEL or NOAEL-Which is correct guidance ICH Q3 or EMA/APIC
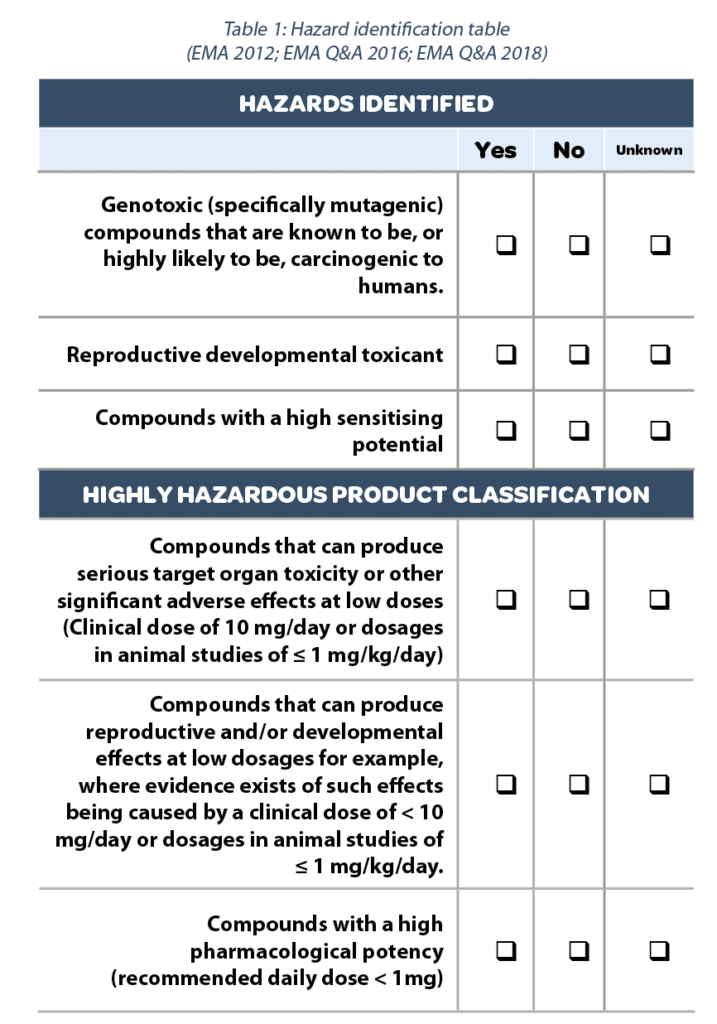
Toxicological approach to define the PDE for your cleaning validation process. - A3P - Industrie Pharmaceutique & Biotechnologie