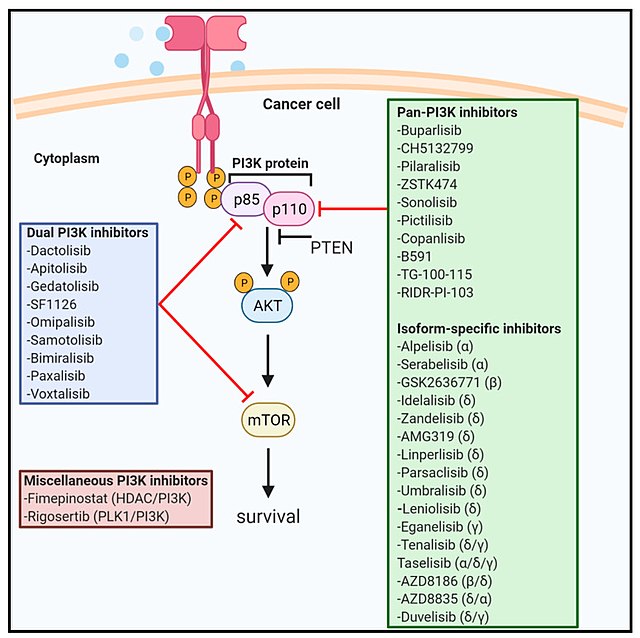
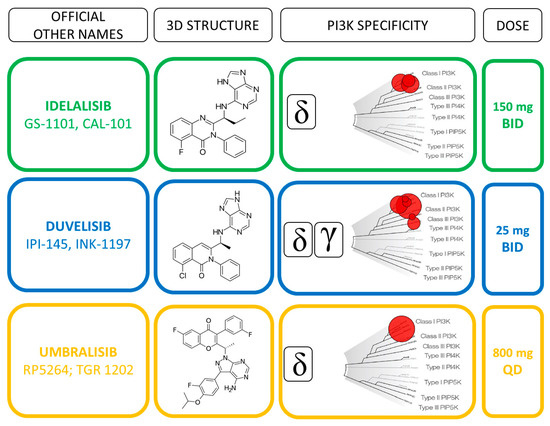
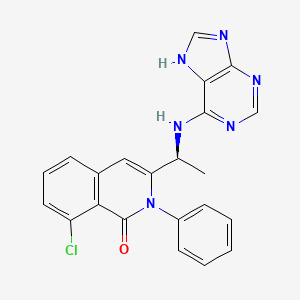
UPDATE_Duvelisib (Copiktra) for the treatment of patients with relapsed or refractory chronic lymphocytic leukaemia (CLL) and re

Michael Hallek on Twitter: "Following the presentations at the 2019 @ASH_hematology meeting and the recent approval of #venetoclax and #obinutuzumab by the European Medicines Agency, we have updated the 1L treatment algorithm
BRIEFING DOCUMENT FOR THE SEPTEMBER 23, 2022, US FDA ONCOLOGIC DRUGS ADVISORY COMMITTEE MEETING COPIKTRA (DUVELISIB) NDA 21115
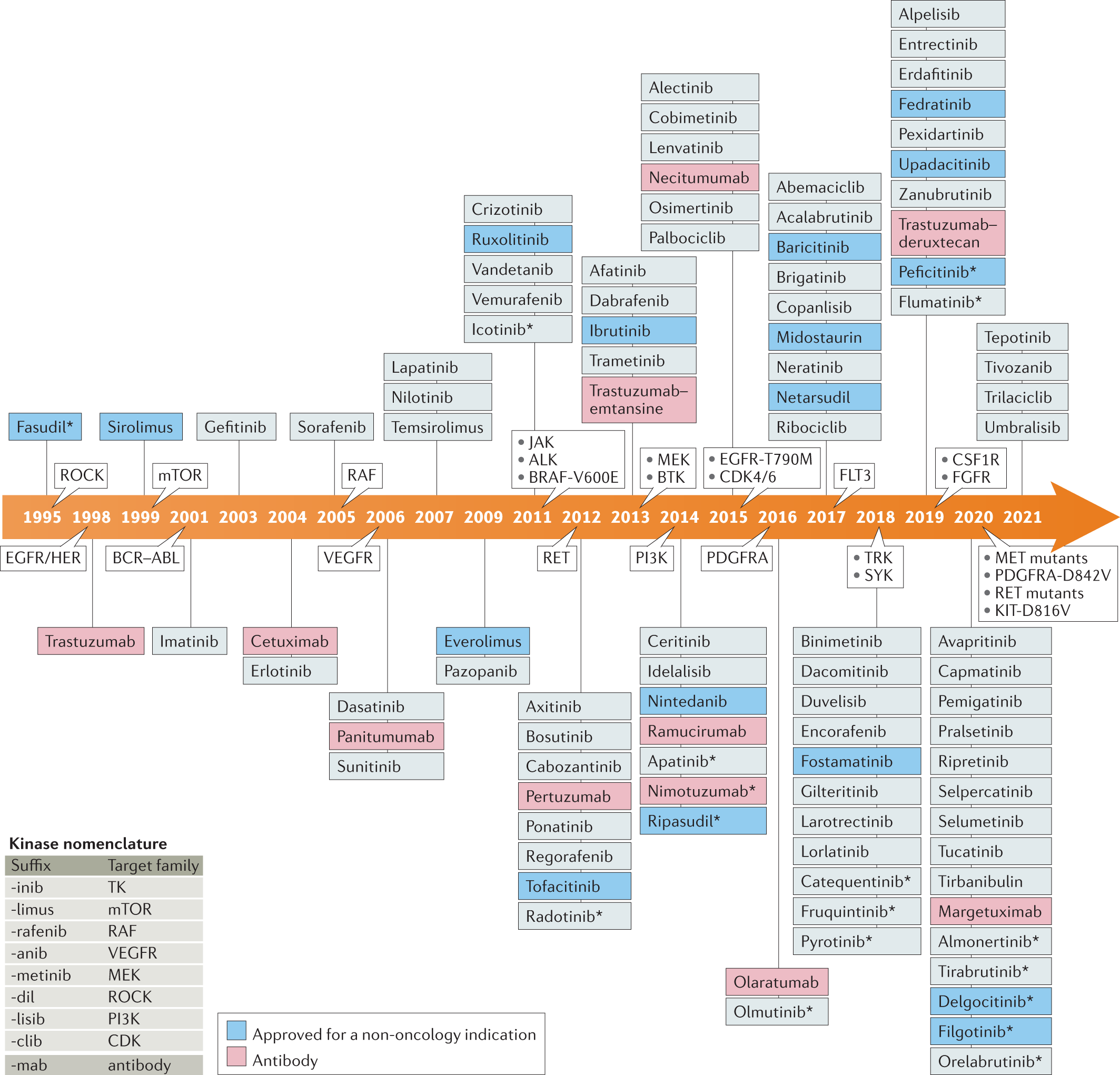
Trends in kinase drug discovery: targets, indications and inhibitor design | Nature Reviews Drug Discovery