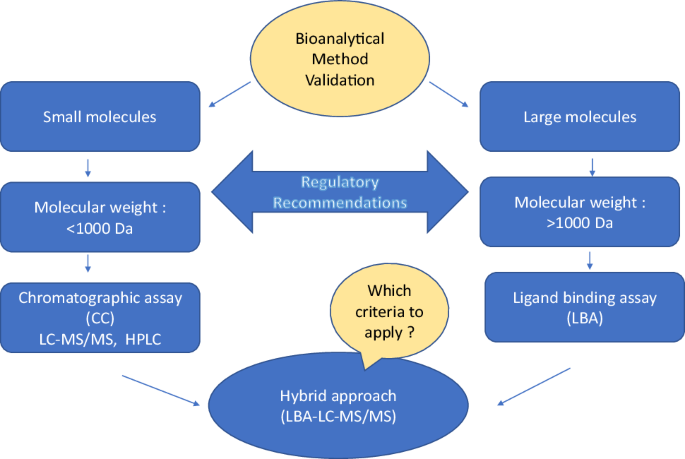
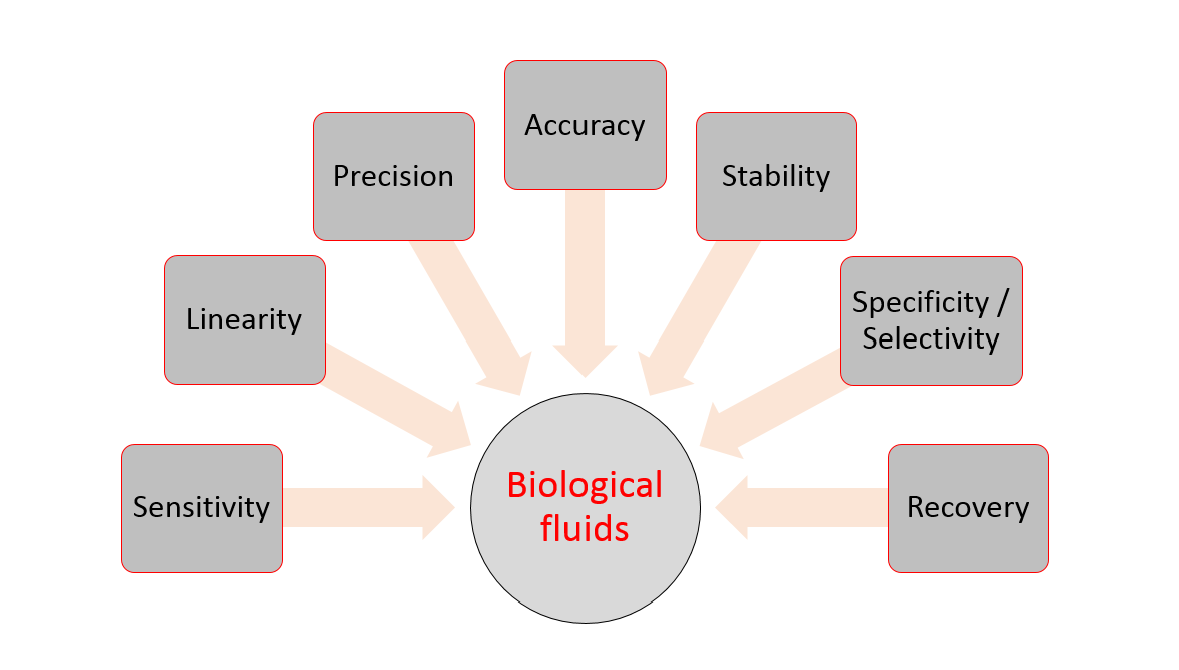
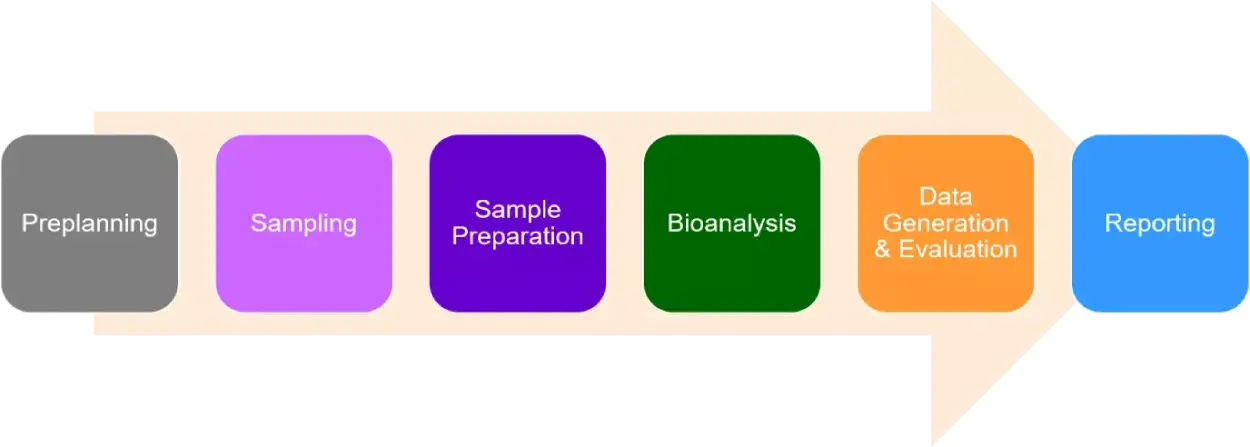
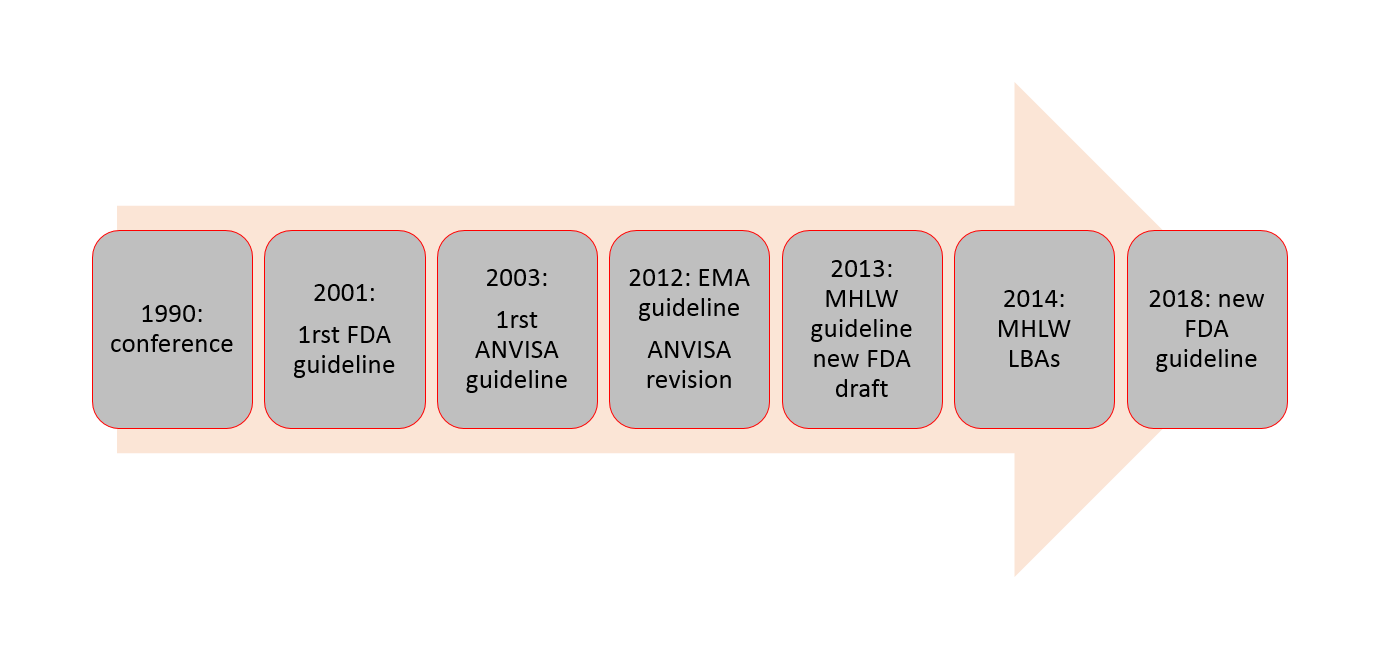
Quantitative evaluation of the matrix effect in bioanalytical methods based on LC–MS: A comparison of two approaches - ScienceDirect

EMA Guideline on immunogenicity assessment of biotechnology-derived therapeutic proteins, December 2007 - Bioanalytical Lab & Top CRO for Bioanalysis

PDF) The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content | Peter van Amsterdam - Academia.edu
![PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ec1710368980e1a3dcbcc921fb7062d9bb753191/4-Table2-1.png)
PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar
![PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ec1710368980e1a3dcbcc921fb7062d9bb753191/6-Table4-1.png)
PDF] omparative assessment of bioanalytical method validation uidelines for pharmaceutical industry | Semantic Scholar

EMA released for public consultation the draft ICH guideline M10 on bioanalytical method validation | EPTRI
The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content.
The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content.