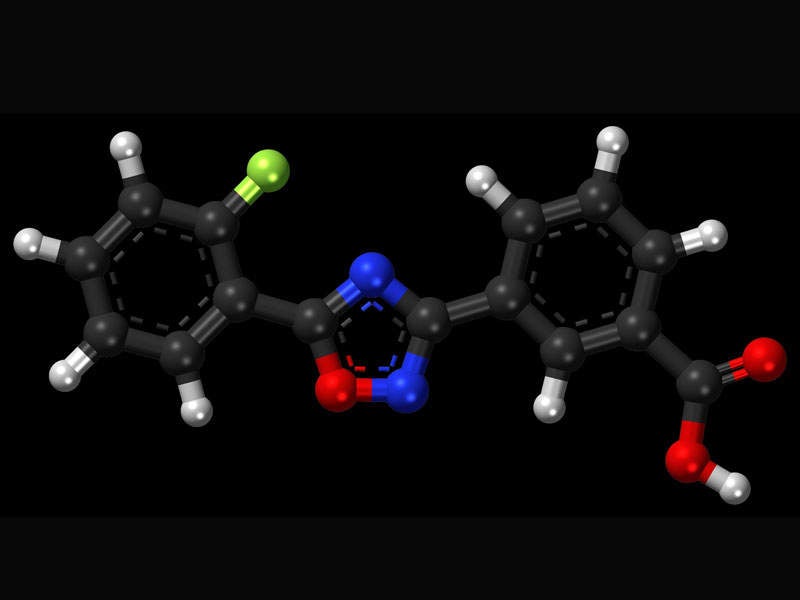
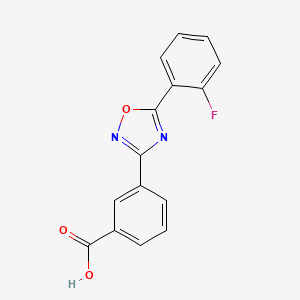
Ataluren use in patients with nonsense mutation Duchenne muscular dystrophy: patient demographics and characteristics from the STRIDE Registry

PTC Therapeutics Announces European Medicines Agency Validation of Marketing Authorization Application for Ataluren in Duchenne Muscular Dystrophy

Andy Biotech on Twitter: "EMA CHMP Jan Meeting Highlights http://t.co/0wgIFR8c6r Negative on $PTCT ataluren, $TEVA $ACTI.ST laquinimod, etc http://t.co/QvuSFipItp" / Twitter
NHS England Enables Access to Translarna™ ▽ (ataluren) For Patients with Nonsense Mutation Duchenne Muscular Dystrophy
EMA Validates PTC Therapeutics' Submission for New Nonsense Mutation Cystic Fibrosis Indication for Translarna (Ataluren)

Clinical trial phase and FDA/ EMA approval status of available gene... | Download Scientific Diagram

EMA reconsiders 'read-through' drug against Duchenne muscular dystrophy following appeal | Nature Biotechnology

Meta-analyses of ataluren randomized controlled trials in nonsense mutation Duchenne muscular dystrophy

Ataluren in patients with nonsense mutation Duchenne muscular dystrophy (ACT DMD): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial - The Lancet
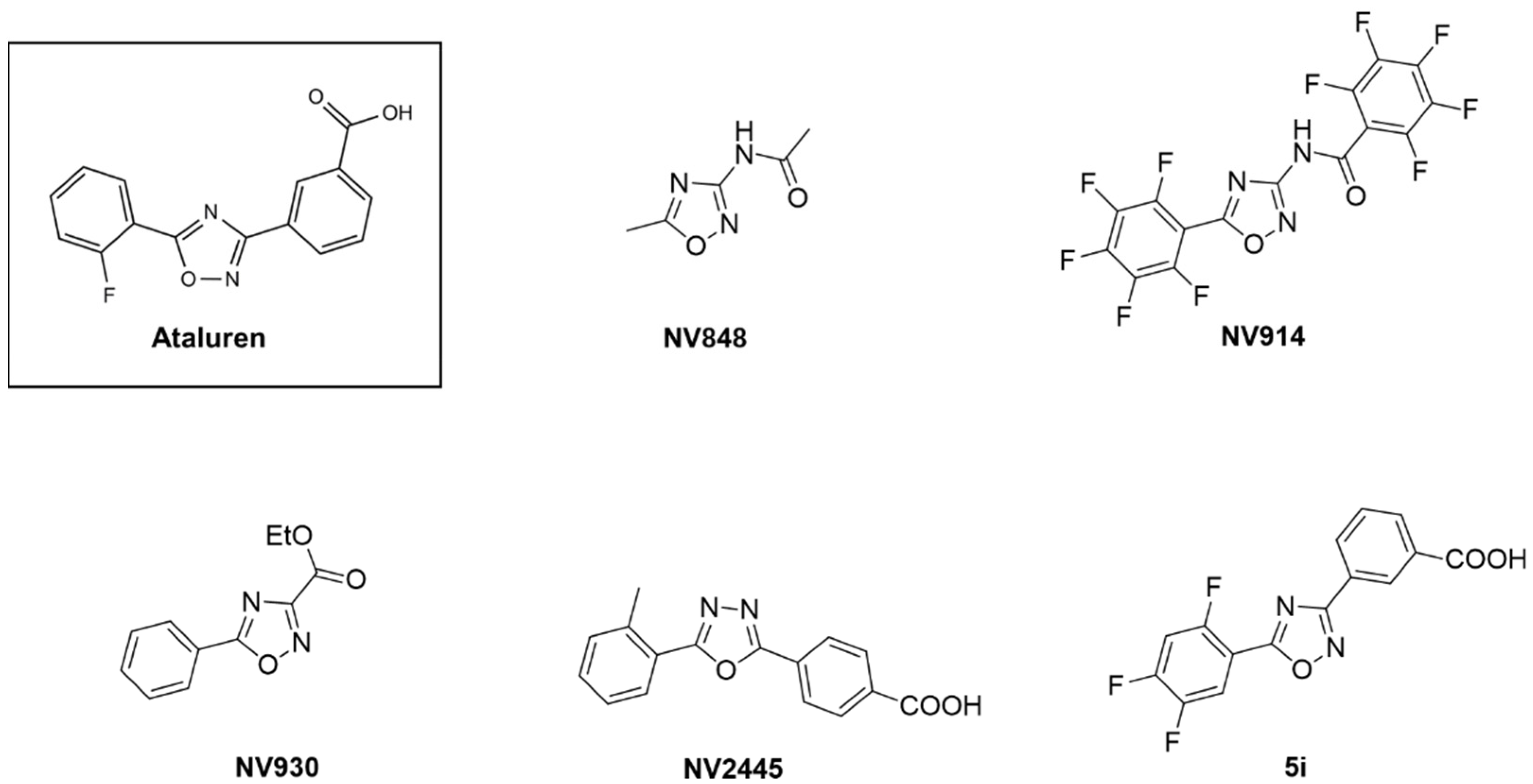
Biomedicines | Free Full-Text | Novel Translational Read-through–Inducing Drugs as a Therapeutic Option for Shwachman-Diamond Syndrome
Ataluren: An Investigational Dystrophin Restoration Drug for Nonsense Mutation Duchenne Muscular Dystrophy

Frontiers | Off-Label Use of Ataluren in Four Non-ambulatory Patients With Duchenne Muscular Dystrophy: Effects on Cardiac and Pulmonary Function and Muscle Strength